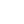Inorganic-Organic Hybrid Materials
Inorganic-Organic Hybrid Materials
Hybrid materials, among them, Metal-Organic Frameworks (MOFs) are coordination polymers consisting of metal ions or clusters linked into extended structures by bridging polydentate organic ligands. These materials are of our special interest and we study several aspects of hybrid materials ranging from their synthesis to applications in different fields. We are also interested in the synthesis of new organic ligands to prepare novel hybrid materials. We are committed to the development of fast, economical and safe routes for the synthesis of hybrid materials. The difficulty lies in the large number of factors that affect the final structure of the product, such as temperature and reaction time, pH, metal/ligand molar ratio, solvent and counter ion type, etc. We are interested in studies of different synthetic routes and parameters to optimize the formation of desired materials. Efforts are given to prepare high-quality single crystals adequate for structural analysis via hydrothermal or solvothermal synthesis routes.
Alternative synthesis methods such as electrochemical, sonochemical and sonoelectrochemical have also been investigated in an effort to reduce the synthesis time and to produce nano-sized hybrid materials.
Selected publications:
- Kulesza, J., Barros, B. S., da Silva, I. M. V., da Silva, G. G., & Alves Júnior, S. Efficient and environmentally friendly electrochemical synthesis of the metallacalixarene [Cu(1,3-bdc)·DMF]·2H2O. CrystEngComm, 15 (2013), 8881-8882. https://doi.org/10.1039/c3ce41679h
- Kulesza, J., Barros, B. S., & Alves, S. (2013). Organic-inorganic hybrid materials: Metallacalixarenes. Synthesis and applications. Coordination Chemistry Reviews, 257 (2013) 2192-2212. https://doi.org/10.1016/j.ccr.2013.03.031
- da Silva, G. G., Silva, C. S., Ribeiro, R. T., Ronconi, C. M., Barros, B. S., Neves, J. L., & Júnior, S. A. Sonoelectrochemical synthesis of metal-organic frameworks. Synthetic Metals, 220 (2016), 369–373. https://doi.org/10.1016/j.synthmet.2016.07.003
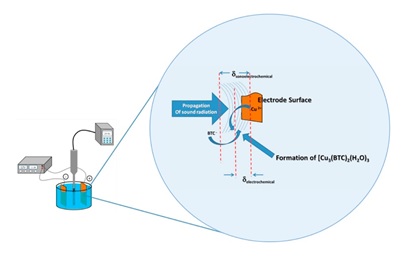
We are also interested in finding structure-property relationships. For example, in the work presented below, we found that three structurally different Zn2+-coordination polymers based on carboxylate and/or benzimidazole ligands can be prepared depending on the type of the counter ion and the temperature used in the synthesis. The thermal and photophysical properties of the two obtained Zn(II) mixed-ligand coordination polymers were dependent on the linkers packing in the solid state.
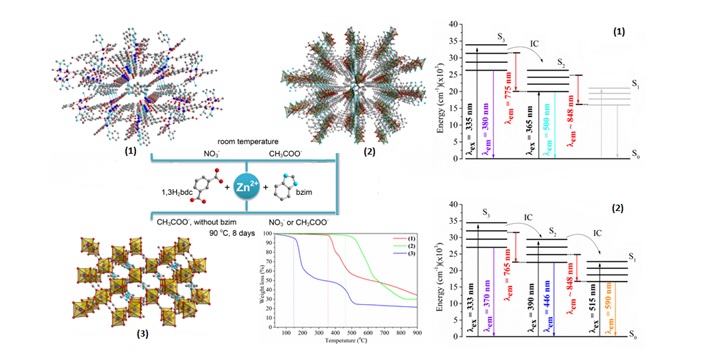
Selected publications:
- Barros, B. S., Chojnacki, J., Macêdo Soares, A. A., Kulesza, J., Lourenço Da Luz, L., & Júnior, S. A. Thermostability and photophysical properties of mixed-ligand carboxylate/benzimidazole Zn(II)-coordination polymers. Materials Chemistry and Physics, 162 (2015), 364–371. https://doi.org/10.1016/j.matchemphys.2015.05.079
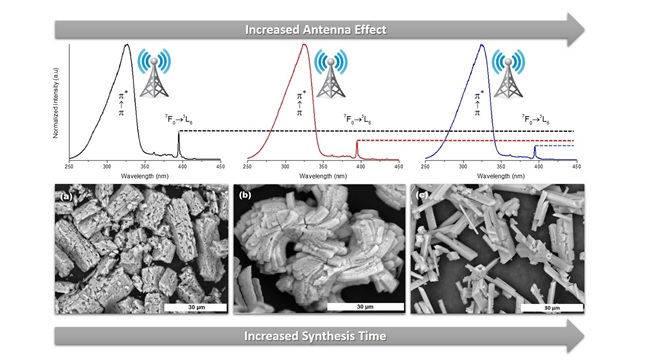
Another central topic of this research is the development of new lanthanide-containing hybrid materials as promising candidates for light-emitting materials, sensors, multimodal image contrast agents and luminescent markers. For example, in the work below, we evaluated the effect of the synthesis time on the microstructure and photophysical properties of Gd/Eu-MOFs. Such systems could be successfully applied as multimodal image contrast agents.
Selected publications:
- do Nascimento, J. F. S., Barros, B. S., Kulesza, J., de Oliveira, J. B. L., Pereira Leite, A. K., & de Oliveira, R. S. Influence of synthesis time on the microstructure and photophysical properties of Gd-MOFs doped with Eu3+. Materials Chemistry and Physics, 190 (2017), 166–174. https://doi.org/10.1016/j.matchemphys.2017.01.024
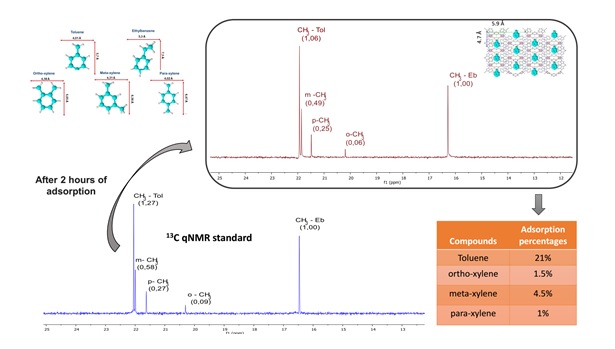
The most recent research line of our group focuses on the application of hybrid materials in the field of healthcare (drug delivery systems) and environmental protection (adsorption of volatile organic compounds and dyes). We mainly use GC and 13C qNMR techniques to quantify the BTEX adsorption in hybrid materials prepared by our research group.